1 Department of Neurosurgery, Great Ormond Street Hospital for Children,
London, United Kingdom; 2 Department of Paediatric Oncology, Alder Hey
Children’s Hospital, Liverpool, United Kingdom; 3 Norton Neuroscience
Institute, Norton Children’s Hospital, Louisville, Kentucky; 4 Department
of Radiology, Alder Hey Children’s Hospital, Liverpool, United Kingdom;
5 Department of Neurosurgery, Alder Hey Children’s Hospital, Liverpool,
United Kingdom; 6 Department of Neuroscience and Neurorehabilitation,
Bambino Gesu Children’s Hospital, Rome, Italy; 7 Department of
Oncology, St. Jude Children’s Research Hospital, Memphis, Tennessee;
8 Neuroscience Center, Texas Children’s Hospital, Houston, Texas;
9 Department of Neurosurgery, Boston Children’s Hospital, Boston,
Massachusetts; 10 Department of Neurosurgery, Nicklaus Children’s
Hospital, Miami, Florida; 11 Department of Neurosurgery, Children’s
National Medical Center, Washington, DC; 12 Neuroscience Institute,
Le Bonheur Children’s Hospital, Memphis, Tennessee; and 13 Semmes
Murphey Clinic, Memphis, Tennessee
Cerebellar mutism syndrome (CMS), or posterior fossa syndrome (PFS), results from surgical manipulation of key nuclei and tracts within the cerebellum and brainstem during resection of a posterior fossa tumor. CMS can range from mild to severe. Since the term “CMS” was first introduced in the 1980s, it has gained awareness within the pediatric neurosurgical and neurooncological communities.
The hallmark feature of CMS is severely reduced or ab- sent speech, typically presenting within the first few days of surgery. However, affected children often experience additional symptoms, including emotional and behavioral lability, cerebellar dysfunction, dysphagia, and other motor signs. In the early postoperative period or acute phase, CMS can manifest as complete mutism or with reduced speech (specifically, an inability to complete a three-word sentence1). In time, paucity of speech gives way to ataxic dysarthria, also known as scanning speech. Although all patients improve with time, the more severe the CMS at onset, the more likely the patient will be afflicted by long-lasting cognitive, motor, linguistic, and social impairments (chronic phase). The incidence of CMS remains high in certain populations, affecting 20%–40% of children with medulloblastoma in one reported series. 2 However, the true incidence is likely higher.
The aim of this paper is to provide guidance for pediatric neurosurgeons on operative strategies and techniques to reduce the incidence of CMS.
Methods
Formed in 2014, the Posterior Fossa Society is an international nonprofit organization of clinicians, researchers, and other allied health professionals who evaluate all aspects of CMS through collaborative working groups. At the April 2024 global meeting of the Posterior Fossa Society, a special session was held on surgical aspects of CMS. Those within the surgical interest group, along with other key members of the Society, came together to craft recommendations on CMS prevention strategies. The recommendations were a result of extensive in-person and remote discussions held during and after the 2024 meeting, supported by a review of peer-reviewed articles andunanimously approved by all authors. All neurosurgeons
were pediatric subspecialty trained with a clinical practice emphasizing neuro-oncology and were thereby well versed in posterior fossa tumor surgery.
Terminology
There have been extensive discussions within the Society with respect to nosology. The term “CMS” was chosen to represent the myriad manifestations of this postsurgical condition, and a definition of CMS was provided by way of a Delphi process undertaken after the inaugural Posterior Fossa Society meeting. 3 Other groups have selected “PFS” as an alternative to “CMS.” At the first open global meeting of the Society in 2020, we decided on “CMS” while agreeing that the term “PFS” was acceptable. Furthermore, some groups have drawn a distinction between postoperative absence of speech as opposed to severely reduced speech. The group at St. Jude Children’s Research Hospital uses the terms “PFS1” and “PFS2” to
differentiate between a postoperative absence of speech versus reduced speech, respectively, while the Nordic European group uses the terms “postoperative speech impairment (POSI) 1 and 2” for the same distinction. 4 In this statement, we use the terms “CMS” and “PFS” interchangeably.
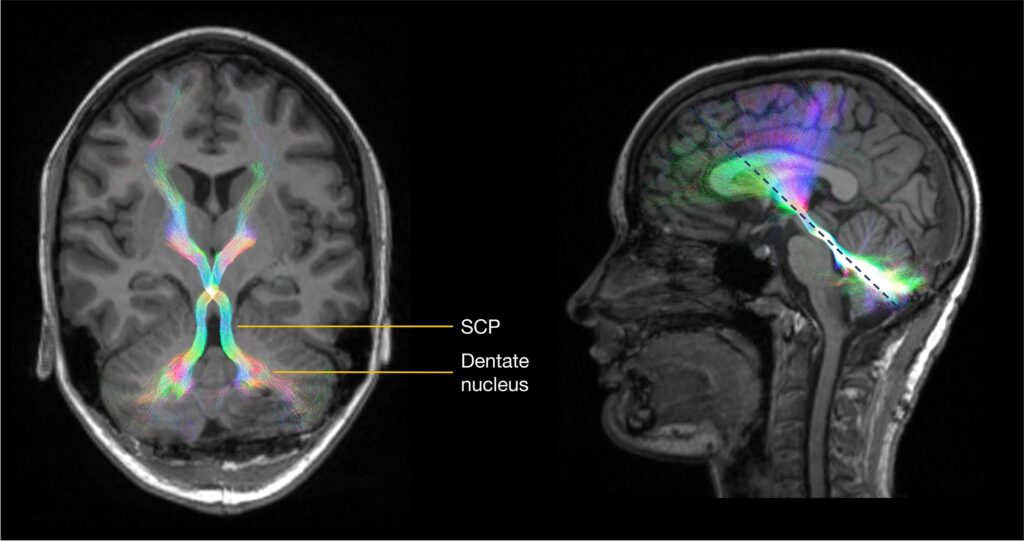
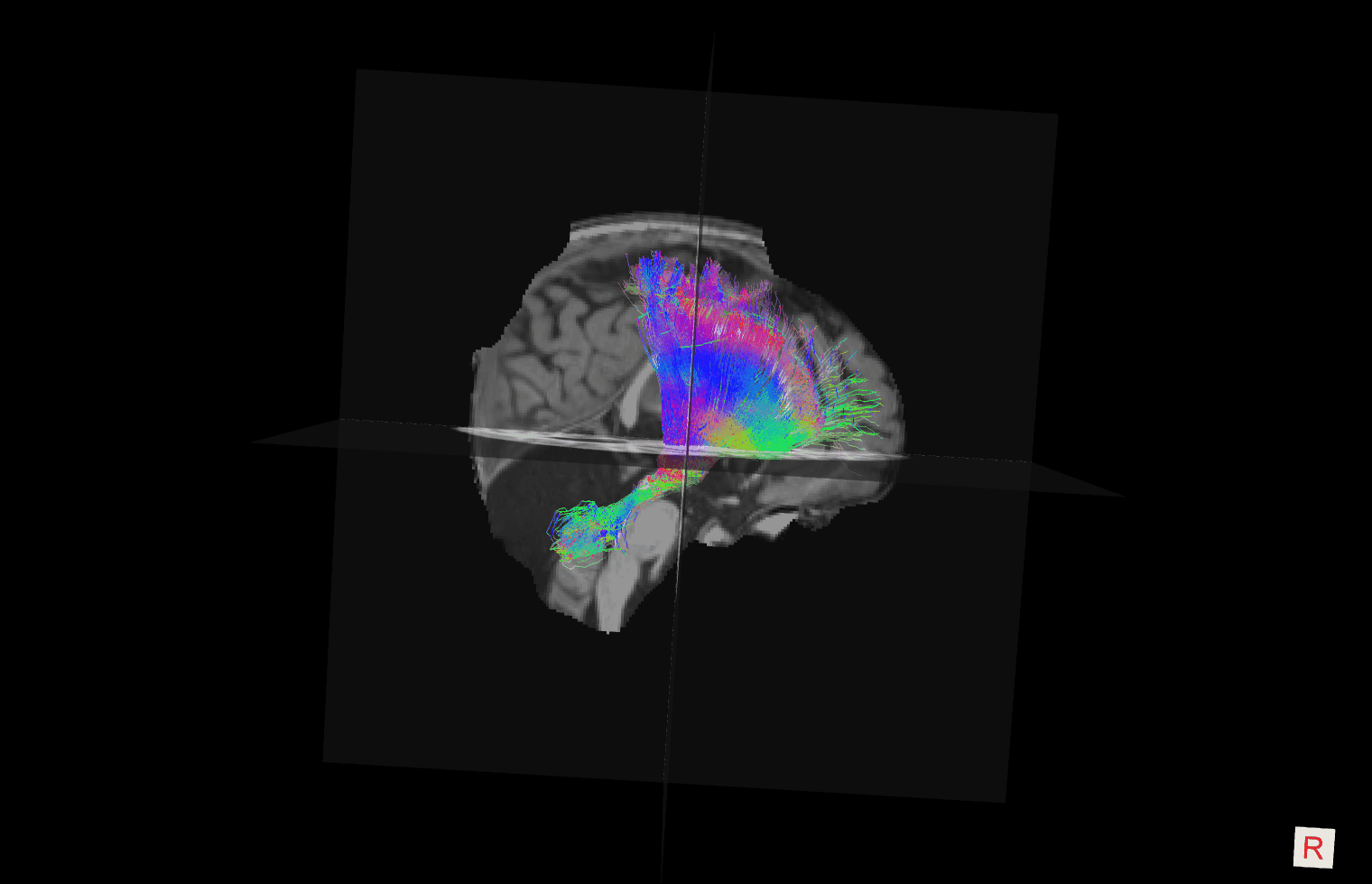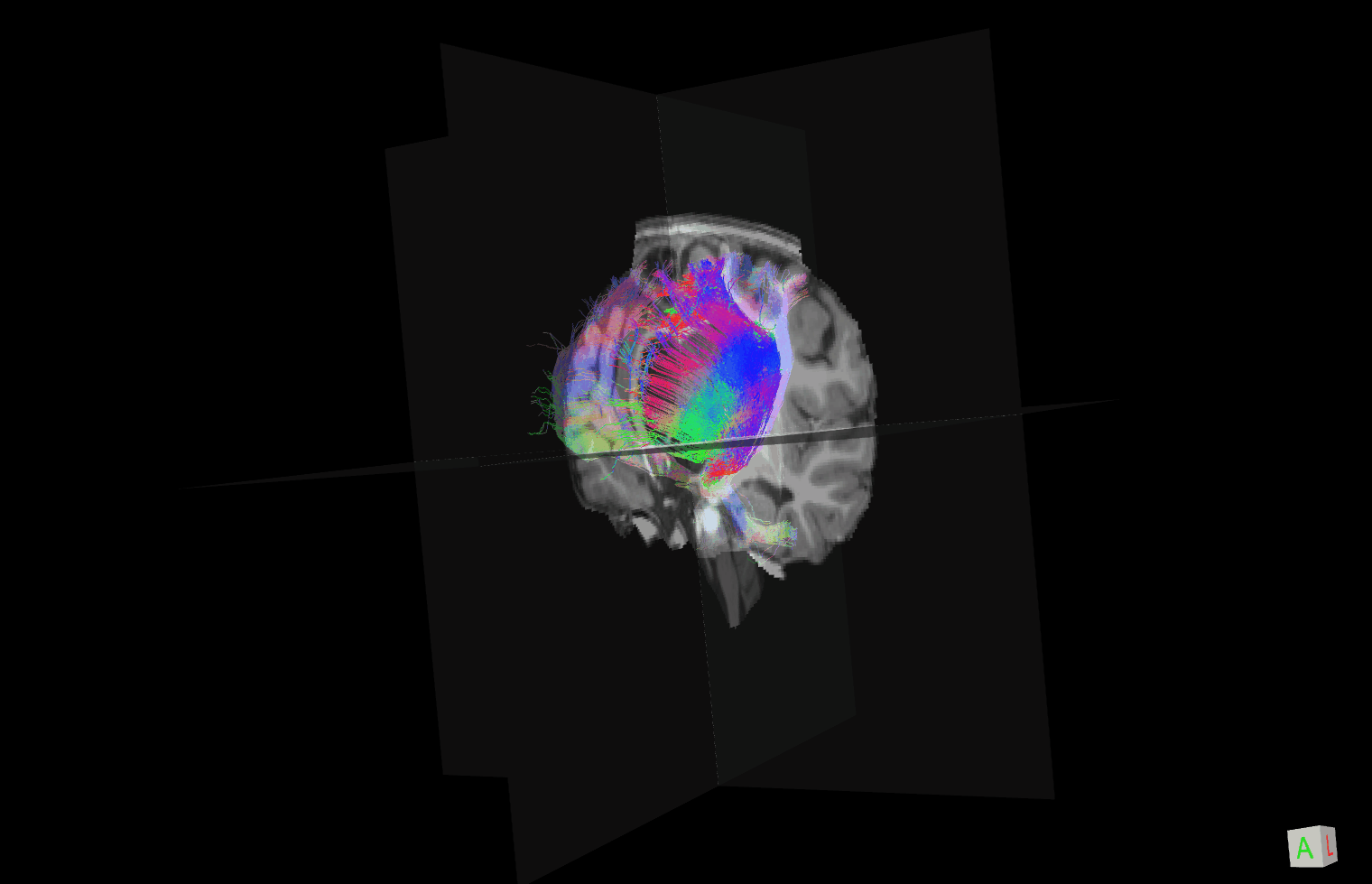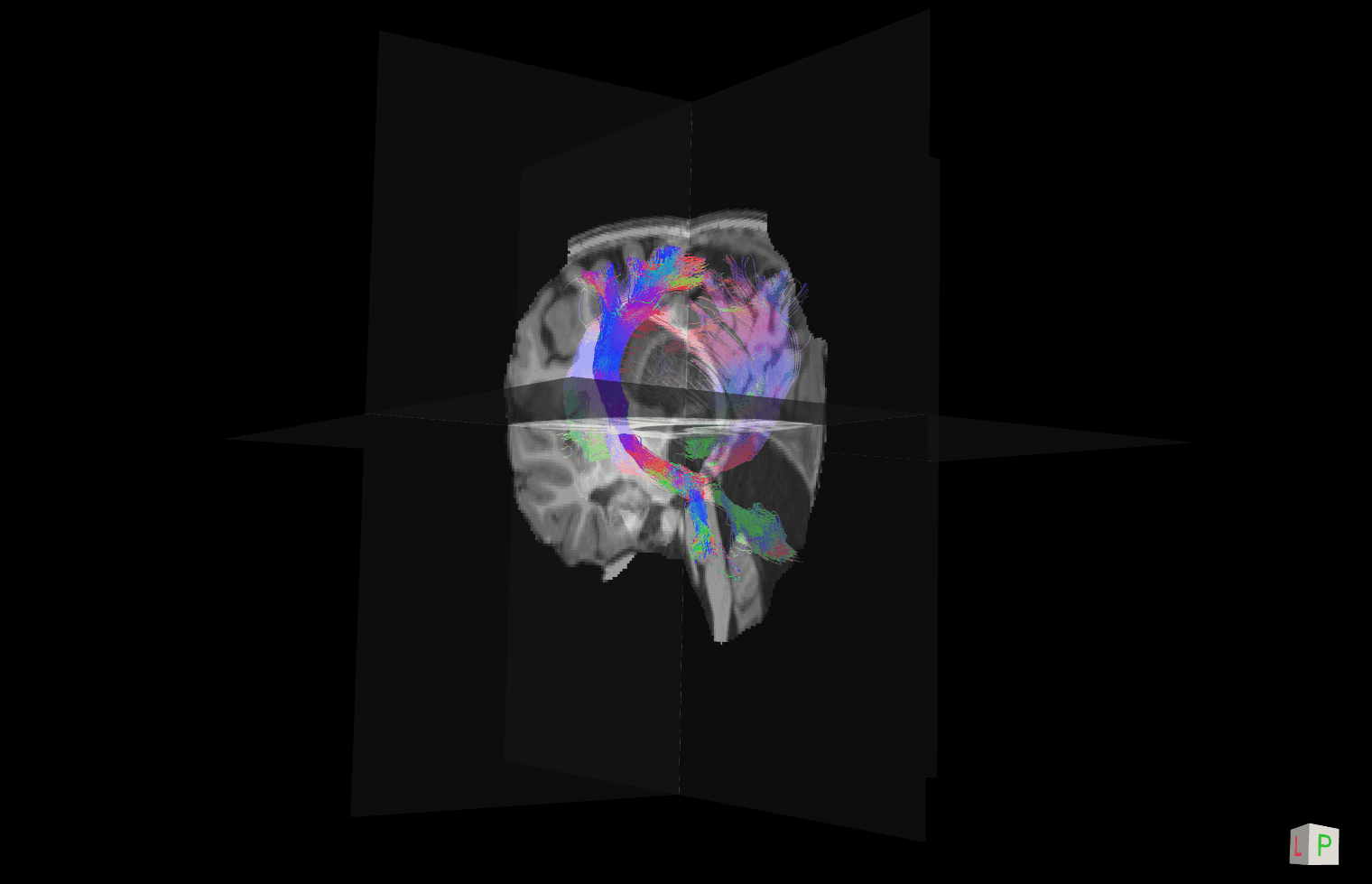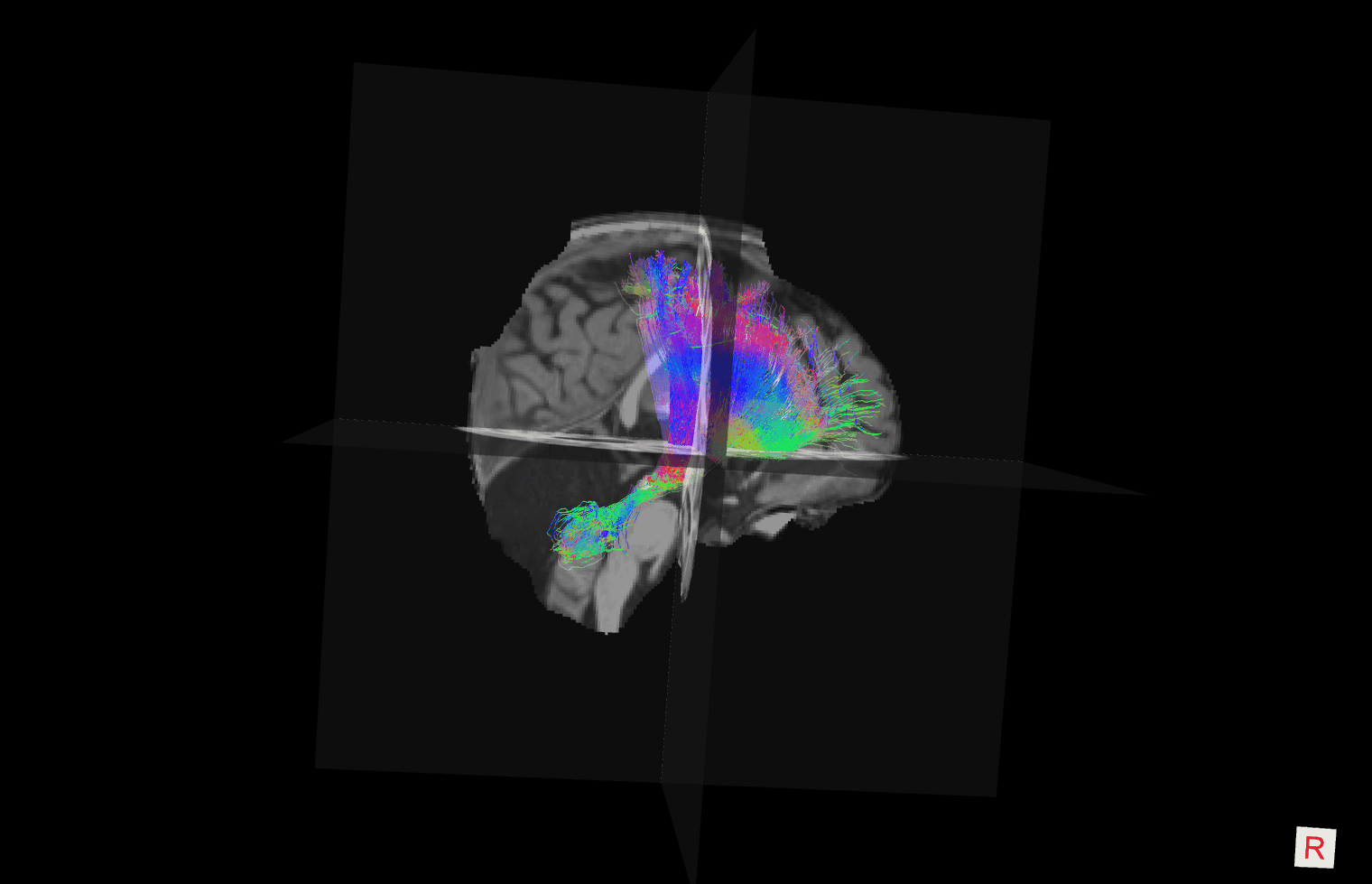
CMS. Bilateral DRTC tract streamlines (in a directionally encoded color scheme) are overlaid on T1-weighted images. Tracts in
their long axis (left) are indicated by the dotted blue line (right). Figure is available in color online only.
Neural Substrates of CMS
It is now widely accepted that disruption of the proximal efferent cerebellar pathway (pECP)—the dentate and other deep cerebellar nuclei, and the proximal dentato-ru-bro-thalamo-cortical (DRTC) tract as it ascends throughthe superior cerebellar peduncles (SCPs) (Fig. 1)—is closely linked to the development of CMS. The cerebellum communicates with other parts of the brain through thecerebellar nuclei. Diffusion signal abnormalities indicating damage to white matter bundles in the SCP were first demonstrated in 2009.5 Similar voxel-based results have been replicated by other groups since <sup6,7 and include dif- fusion abnormalities of the pECP on intraoperative MRI scans, which were significantly associated with CMS,with bilateral involvement of pECP being highly specific.7 Hypertrophic inferior olivary degeneration (by way of the central tegmental tract) is another characteristic imaging feature in patients with CMS (Fig. 2).
8 Recent studies have also identified damage to the fast-igial nucleus as a strong predictor of CMS.9 Diffusion tractography has revealed the underlying somatotopy of the SCP in children10 and shown that SCP damage in CMS may be highly focal.11 These diffusion imaging studies are complemented by modern lesion-symptom mapping approaches that demonstrate higher lesion loads in the cerebellar outflow pathway in children with CMS compared to those without.12,13
While bilateral damage to the pECP structures has been significantly associated with CMS, a larger number of studies have demonstrated a greater risk of CMS associated with injury to the right side of the cerebellum and pECP compared with the left side. Ji et al. showed that unilateral damage to the DRTC tract increased the odds by 14.4 (absent right DRTC tract) and 8.5 (absent left DRTC tract), and these patients had a less severe CMS (i.e., PFS2/POSI2).14 The Rotterdam predictive model for CMS assigns twice as many points to patients with right-sided middle or SCP involvement or invasion.15 This elevated risk with right-sided cerebellar involvement is thought to be due to the projection of the DRTC tract to the left side of the cerebrum, which more commonly harbors language centers and their interconnectivity. Perfusion imaging techniques have shown alterations in supratentorial blood flow in children with CMS, mainly affecting the frontal lobes.16
Risk Factors
Established nonmodifiable risk factors include tumor location, size, and type as well as age.17,18 Large midline
tumors, most significantly intraventricular (vs intracerebellar) tumors, that splay or even invade the cerebellar peduncles are the most important risk factor (Figs. 2 and 3). While CMS can occur with any tumor, medulloblastoma carries the highest probability. In the era of molecular characterization of medulloblastoma, WNT and groups 3/4 are significantly more likely to be associated with CMS than SHH subtype tumors because the latter is more commonly found within the cerebellar hemisphere versuthe midline.1,19 Finally, young children (< 5–10 years of
age) are more likely to develop CMS,1,19 as well as those with preoperative language deficits.20 Such risk factors are included in the various imaging feature-led preoperative risk stratification models,15,21,22 which are currently undergoing multicenter validation. There are several other potential risk factors that have not been consistently borne out in the literature, including sex (males may be at higher